When teaching SN1 and SN2 reactions to my students, this famously difficult duo is made perfectly manageable by breaking down their mechanisms side by side. I always explain to my students that these concepts can be learned and understood much like any of history’s great pairs. Like Tom & Jerry, Tweedle Dee & Tweedle Dum, or Bert & Ernie, these two reaction types make the most sense when they’re together. And my students agree. Comparing key parameters of the two reaction pathways allows the students to clearly see the differences between the SN1 and SN2 reactions, and thus, understand each with clarity.
I begin my lesson by comparing the most critical parts of the two nucleophilic reactions and conclude with the more ancillary details that help in identifying one over the other. My goal is to make this challenging subject matter as clear as possible, and I outline the parameters in the following order:
1. Reaction center of the electrophile
2. Nucleophile type
3. Product stereochemistry
4. Solvent types
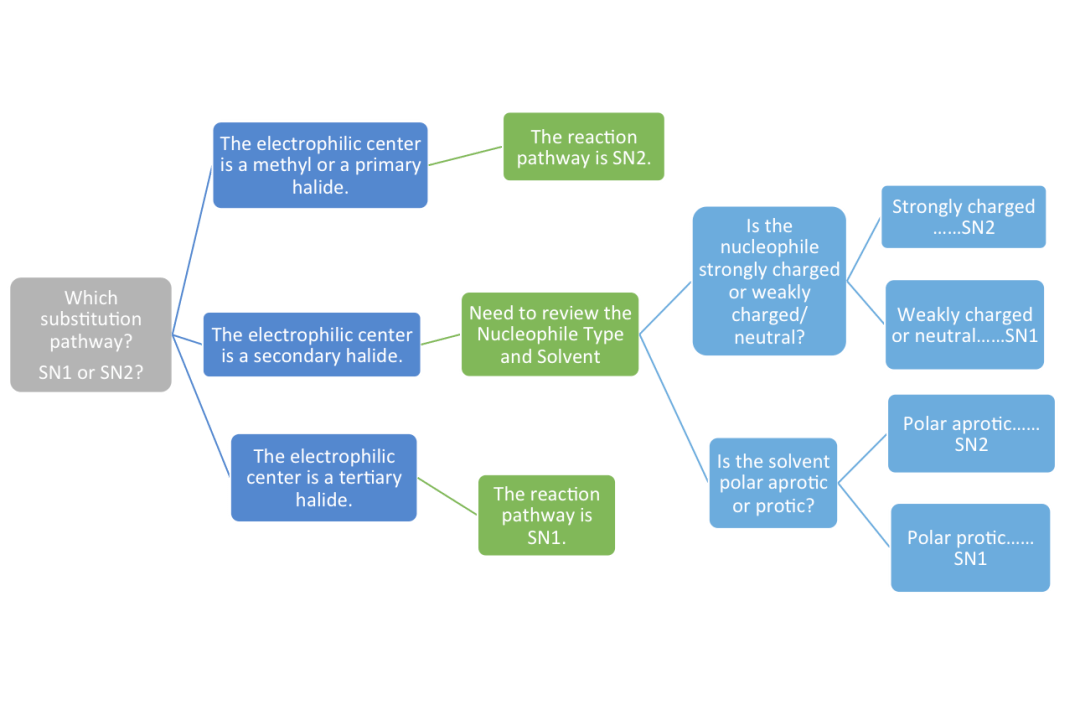
The reaction center of the electrophile can be quite revealing with regards to the reactivity and specificity of the SN1 versus the SN2 reactions. Identifying the carbon center bound to the leaving group is half the challenge in making sense of the difference between the two pathways. By distinguishing the electrophilic center as a methyl, primary, or tertiary halide, the students can contextually separate the SN2 from the SN1 reactions. This leaves the student to tackle identifying a secondary electrophilic reaction center.
For further understanding, my lesson is geared toward clarifying and contrasting the nucleophilic types and product stereochemistry particular to SN1 and SN2 reactions. Multiple examples based on the same secondary starting material, but with varied reactivity and charges, aid student comprehension and serve to give contrast to the two reactions. Lastly, I conclude the lesson with a brief discussion of the solvents specific to the SN1 and SN2 reactions. I always emphasize that this factor can serve as supporting evidence when trying to identify the reaction type.
The mechanisms of the SN1 and SN2 reaction can be clearly compared and contrasted when shown with a secondary electrophilic halide. The secondary halide is usually the source of a lot of confusion. Thus, keeping the starting material the same and looking at the mechanism of two vastly different nucleophiles helps to differentiate the two nucleophilic reactions (See Figure 1).

As shown with Figure 1, the comparison method of the reaction pathway and mechanism can help show the specificity of the products and pathway. Mechanistically, the students can see that the SN2 reaction is concerted through backside attack and the SN1 reaction is step-wise – allowing rearrangements and multiple products. Getting down to the mechanisms really communicates the fundamental differences between these pathways. What is complicated and abstruse from a functional perspective is made completely clear when viewed mechanistically. Thanks to this approach, my students are better acquainted with this illustrious pairing than ever before.
-Kerri Shelton Taylor, Columbus State University
Click here to learn more about Professor Taylor
I like the flow chart, is that something that you incorporate into your lecture/class directly? Seeing that I thought it might make an interesting homework assignment to ask students to come up with their own chart to tell between the two (or even including E1/E2)