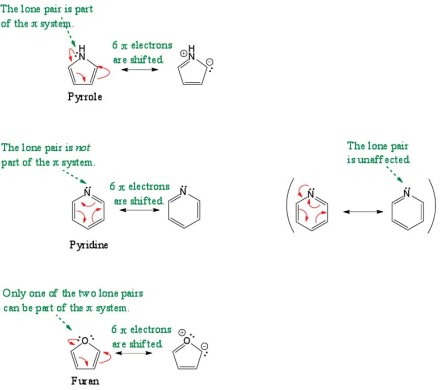
One of the things my students find most challenging about aromaticity is whether to include lone pairs as part of a cyclic π system. If a lone pair is included, then the number of π electrons increases by two, and a student’s prediction about whether a species is aromatic will also change. What I think makes this challenging is that the rules appear to change depending on the nature of the ring and the nature of the atom that has the lone pair. This issue is apparent when we compare pyridine, pyrrole, and furan:
Pyridine and pyrrole both contain a nitrogen atom with a lone pair of electrons, but, whereas the lone pair in pyridine is not included in the π system, the one in pyrrole is. In furan, one of the lone pairs on oxygen is included but not the other.
Students can certainly memorize these specific results for pyridine, pyrrole, and furan, but the true problem manifests itself when students are asked to make predictions about the aromaticity of unfamiliar molecules that contain atoms with lone pairs. For many years my students struggled with this, but with an additional tool I now teach, they have become quite good at making these kinds of predictions.
Years ago I would explain the results of pyridine, pyrrole, and furan just as textbooks do, using only arguments that invoke orbitals, as follows:
Over the years, however, I saw that this explanation benefited only my best students, leaving the others behind. I think one reason is that, to use these orbital arguments, students must envision orbitals in the Lewis structures or line structures they are typically given in a problem, something that many students have difficulty doing. Second, it requires students to consider various hybridizations for the atom with the lone pair—the hybridization that students would normally predict via VSEPR theory and one that might benefit the molecule by resulting in aromaticity.
I still teach the orbital arguments as presented above, but now I also show students how to arrive at the same answers using only resonance theory, something that students are already very comfortable with by this point in the course. The idea is that the atoms over which electrons can be shifted via resonance are the ones that contribute p orbitals to the same π system, and the number of curved arrows that are used to shift the electrons corresponds to the number of pairs of electrons in that π system.
In pyrrole, six electrons can be shifted via resonance, involving all five atoms of the ring, so there is a single, cyclic π system containing six π electrons. In pyridine, the double bonds are shifted via resonance without involving the lone pair (as shown above on the left), so the lone pair is not part of the same π system as the double bonds. (On the right above, I show that an attempt can be made to involve that lone pair in resonance, but doing so results in a resonance structure in which the lone pair remains on N.) Finally, for furan, just one of the lone pairs on oxygen is involved in resonance with the double bonds, so the second lone pair is not part of that cyclic π system. (Simultaneously including the second lone pair on oxygen in resonance would exceed an octet on one of the atoms.)
Now that I teach both methods of predicting whether a lone pair is part of a π system—one that invokes arguments of orbitals and one that circumvents orbitals—I find that I am reaching many more students. For this reason, I have included both methods in my textbook in the chapter on conjugation and aromaticity (Chapter 14). My better students still appreciate the depth of understanding they receive with an orbital explanation, while all of my students are better equipped to solve problems on aromaticity.
I would be interested in hearing from others about how they and their students deal with these types of problems.
— Joel Karty


That helps me………….Thanks
Thank You Sir….the description u provide helped
This is what my teacher taught me long ago..but i forgot as the time passed..thanks to you which helped me to remember..
“In Pyridine , six electrons can be shifted via resonance, involving all five atoms of the ring, so there is a single, cyclic π system containing six π electrons. In pyridine, the double bonds are shifted via resonance without involving the lone pair (as shown above on the left), so the lone pair is not part of the same π system as the double bonds. (On the right above, I show that an attempt can be made to involve that lone pair in resonance, but doing so results in a resonance structure in which the lone pair remains on N.) Finally, for furan, just one of the lone pairs on oxygen is involved in resonance with the double bonds, so the second lone pair is not part of that cyclic π system. (Simultaneously including the second lone pair on oxygen in resonance would exceed an octet on one of the atoms.)”
I think you wanted to say “In pyrrole” rather “In pyridine”? In the first line of the quotation
And Thanks so much for the explanation, it helped me as well.
Do you know why true scientists are great people? One of the reasons is that they keep accepting small notices or remarks from even a student with little knowledge of chemistry (like me). I think you are a true scientist and a great person, Thanks.
thanks a lot!!! 😘
Best concept….. It will surely help me to solve this type of problems
I like the way you explain this point. My question is close to this issue. As a teacher of General Chemistry, I have problems to explain why In pyrrole and furano, if the stochiometric formula according to Lewis’ theory is AX3E (N) and AX2E2 (O), the electronic geometry is not tetrahedral but planar trigonal. The concept of aromaticity should not be invoked at this very moment. I hope you understand my worry.
Thanks in advance.
J. Manuel Recio
University of Oviedo
Oviedo, Spain
Lovely explanation! Helped me out!
Very nice concept.
Which one has higher resonance energy
nice explanation
This was awesome! I am teaching this tomorrow! Thanks!
Thank you sir…the explanation you provide it helped to me to solve this kind of questions..
Its indeed a very nice method.
Its indeed a very nice method. Thankyou
this makes sense but it did not clear my doubt. I mean, by what you have said we can make out how many lone pairs get into the ring provided the arrow are given. What if only the bond structure is given?? And as it is an unfamiliar molecue we do not know in which orbitals the lone pair is in??
then what will we do? how will we know?? i hope you understood my doubt.
Thankyou
i keep it super simple and say — if the lone pair electrons are needed to complete the (4n+2) electron rule, then they are; otherwise, they are not.